
Prof. Deng Cheng’s team, from Center for
High Altitude Medicine, West China Hospital, Sichuan University (SCU),
published in Nature an article titled "Constitutively Active Glucagon
Receptor Drives High Blood Glucose in Birds". From the perspectives of
molecular evolution and physiological adaptation, this research innovatively
proposes the perpetual motion molecular model of avian GCGR (glucagon
receptor), revealing how constitutively active GCGR involved in glucose-lipid
and energy metabolic adaptations in birds.
Blood glucose levels in animals are
primarily regulated by the glucagon receptor (GCGR) family, which maintains
high sequence similarity and functional conservation across vertebrates. While
birds and reptiles share similar glucose-lipid metabolic processes with mammals
(including humans), they exhibit significant evolutionary differences.
In vertebrates, carbohydrate homeostasis is
critical for energy requirements and overall health. Blood glucose levels are
tightly regulated within metabolic homeostasis and maintained through
species-specific feedback mechanisms. However, this homeostasis is disrupted in
birds. As early as 1893, German physician Minkowski and colleagues observed
that birds have significantly higher blood glucose levels than other
vertebrates. However, for over a century, the molecular mechanisms underlying
this hyperglycemia have remained an unsolved scientific mystery.
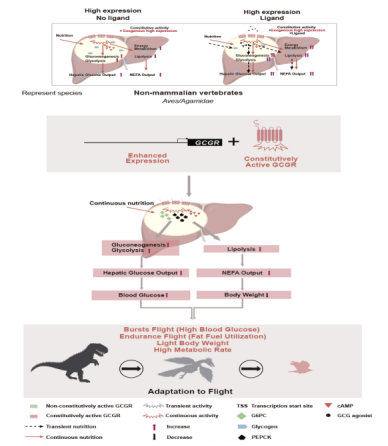
Through molecular evolution analyses and
constitutive activity screening of GCGR across vertebrates, researchers
discovered that avian GCGR exhibits high constitutive activity and is highly
expressed in the liver. Among vertebrates, placental mammals lack constitutive
GCGR activity but exhibit high hepatic expression levels. Non-placental mammals
have weak constitutive GCGR activity and high hepatic level. Most non-mammalian
vertebrates express GCGR at low levels in the liver but possess strong constitutive
activity. Only birds and Agamidae lizards (e.g., bearded dragons) have high
hepatic expression of constitutively active GCGR.
The study also identified eSNPsin promoter
regions that potentially regulate GCGR gene expression.
The team conducted cellular experiments and
in vivo studies across multiple vertebrate species, including zebrafish,
bearded dragons, leopard geckos, budgerigars, chickens, white-rumped munia and
mice. They employed AAV-based gene delivery and genetic editing to confirm that
both overexpression and knockdown of constitutively active GCGR alter blood
glucose levels in different vertebrates. Additionally, the study identified a
naturally occurring point mutation (hsGCGRH339R) in the ICL3 region of the human
GCGR, which exhibits weak constitutive activity. When overexpressed in the
liver of mice, this mutation resulted in hyperglycemia and weight loss.
This research significantly advances our
understanding of adaptive evolution in biological systems. The team proposed
that glucose, as the primary short-term fuel for avian flight, enhances rapid
carbohydrate energy supply during takeoff, enabling flight bursts. The
GCGR-driven hyperglycemia in birds provides a large energy reservoir for
sustained flight. Birds utilize this constitutively active GCGR-mediated energy
supply to support flight while evolving additional strategies such as migration
to adapt to environmental changes.
From a molecular evolution and
physiological adaptation perspective, the study also deciphers the downstream
mechanisms of constitutively active GCGR, providing critical insights into
metabolic homeostasis. These findings may offer theoretical foundations and
potential therapeutic targets for treating diabetes, obesity, and metabolic
liver diseases.
Zhang Chang and Xiang Xiangying, two Ph.D.
candidates from West China Hospital, are the co-first authors of the paper,
with Prof. Deng Cheng as the corresponding author.
Paper link: https://www.nature.com/articles/s41586-025-08811-8